IMAGE CREDIT/OBTAINED FROM: https://mappingignorance.org/2015/12/17/einstein-and-quantum-solids/
Quite some time ago, I had posted a numerical study of an Einstein solid and I now present the analytical study of an Einstein solid. As this was one problem in one of my problem sets while studying thermodynamics and statistical mechanics, one may find this exact problem in the following text:
Schroeder D.V., An Introduction to Thermal Physics, (2000). Addison Wesley Longman. Chapter 3: Interactions and Implications: Problem 3.25. pp. 108.
What I will be presenting is my solution to this problem and I will be offering my interpretation of the problem statement and the implications of the solution.
We begin with the provided approximation,
This expression represents the multiplicity of an Einstein solid with oscillators and
energy units. Recall that the equation to find the entropy is the following
Thus, upon substitution of the multiplicity (Eq.(1)) into the equation for entropy (Eq.(2)), one arrives at the equation
Upon making use of the properties of logarithms, we may write the equation equivalently as
and again using the well-known property that , we may further simplify Eq.(4) such that we arrive at the expression for the entropy of an Einstein solid:
We may omit the factor of from Stirling’s approximation owing to the fact that if
and
are very large numbers, then
and
. Hence, it follows that
. So we see that the aforementioned factor is of no consequence provided that
and
(i.e. the number of energy units and oscillators) is very large.
The second part of this problem asks to take the expression we derived for the entropy and compute the temperature. Recall that the definition for temperature is given by the equation
From substitution we may write the following
where I have made use of the properties of logarithms and made the substitution . Differentiating and simplifying yields,
Further simplification yields the equation for temperature of an Einstein solid
Part three asks us to find the equation for the heat capacity from our temperature equation. Recall that the equation for heat capacity is of the form
However, we need an equation for the internal energy as a function of temperature
. We actually have the opposite. So we must solve Eq.(9) for the internal energy, and doing so yields
Substituting Eq.(11) into Eq.(10) gives
Using the quotient rule for derivatives gives the equation for the heat capacity
The next part asked to show that in the limit , the heat capacity
. Recall that the Taylor series expansion for the exponential function
is given by
For small values of we may make the approximation
. Then we have the approximate relation
Then considering the aforementioned limit yields
The final part of my solution to this problem (I did not complete the last portion of the problem see the aforementioned reference for the problem) asks us to graph the resultant equation relating the heat capacity and the temperature. Below is a plot of the function in the technical computing software Maple.
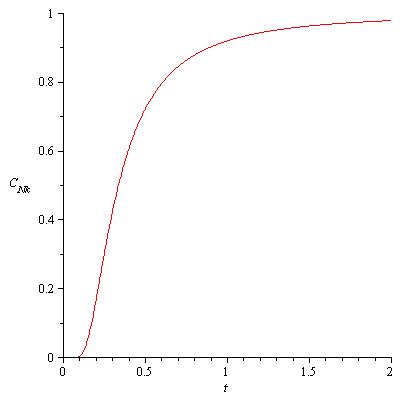
Fig.1 Heat Capacity vs. Temperature
For low temperatures, the heat capacity initially starts at . However, when
, there appears to be a dramatic increase in the heat capacity in the dimensionless quantity
. If the heat capacity
is graphed as a function of temperature
, and one uses the
values for each of the lead and aluminum curves produced in Fig. 1.14 of Schroeder’s An Introduction to Thermal Physics.
Let me know if I made any mistakes anywhere, and I will do my best to correct them.
Clear skies!
