Every physics major at some point in their undergraduate career takes a course in thermodynamics and statistical mechanics. One of my problem sets included a problem that considers an Einstein solid with 50 oscillators and 100 units of energy and then increases the number of oscillators to 5000. I will be presenting my solution to the numerical side of the problem. An Einstein solid can be regarded as
“… a collection of microscopic systems which can store any number of energy ‘units’ of equal size which occur for any quantum-mechanical harmonic oscillator whose potential energy function has the form
…The model of a solid as a collection of identical oscillators with quantized energy units…”
described (defined) by Schroeder in his text Introduction to Thermal Physics. Figure 1 represents the Einstein solid as a whole (in a lattice) and Figure 2 depicts the quantum-mechanical harmonic oscillator interpretation of an Einstein solid.
The problem statement is:
“Use a computer to study the entropy, temperature, and heat capacity of an Einstein solid, as follows. Let the solid contain 50 oscillators (initially), and from 0 to 100 units of energy. Make a table, analogous to Table 3.2, in which each row represents a different value for the energy…Make a graph of entropy vs. energy, and a graph of the heat capacity vs. temperature. Then change the number of oscillators to 5000, and again make a graph of the heat capacity and temperature and entropy and energy, and discuss the predictions and compare it to the predictions to the data for lead, aluminum, and diamond. Estimate the numerical value of
for each of those solids.”
This problem can be found in the aforementioned text.
Figure 1. Einstein Solid (Lattice); Image Credit/Obtained from https://mappingignorance.org/2015/12/17/einstein-and-quantum-solids/
Figure 2. Quantum-Mechanical Harmonic Oscillator interpretation of an Einstein solid as a collection of these oscillators. Image Credit: http://hyperphysics.phy-astr.gsu.edu/hbase/Therm/einsol.html
Part I: Let units, and let $N = 50$. The corresponding data table for this Einstein solid follows. The following set of equations were used to determine the multiplicity and entropy.
and
where is the multiplicity. The remaining quantities of temperature were obtained using a simplified form of the central difference equations for the first order derivative. The respective definitions of temperature and heat capacity are
and
where represents the internal energy of the Einstein solid, and
is the entropy. The generalized from of the first order central difference approximation has the form
where represents the higher order terms, in this case, the quadratic, cubic, quartic, and so on, and
is the step size for each iteration. For the final iteration (when
units), instead of using the central difference approximation, a backward difference approximation was employed since there does not exist data for
units of energy. The backward difference approximation has the form
Table I (Dimensionless Parameters):
| Energy q | Ω | S/k | kT/ε | C/Nk |
| 0 | 1 | 0 | 0 | N/A |
| 1 | 50 | 3.912023005 | 0.27969284 | 0.121826198 |
| 2 | 1275 | 7.150701458 | 0.328336604 | 0.453606383 |
| 3 | 22100 | 10.00333289 | 0.367875021 | 0.536183525 |
| 4 | 292825 | 12.58733044 | 0.402937926 | 0.593741905 |
| 5 | 3162510 | 14.96687657 | 0.43524436 | 0.637773801 |
| 6 | 28989675 | 17.18245029 | 0.465656087 | 0.673043377 |
| 7 | 231917400 | 19.26189183 | 0.494675894 | 0.702124659 |
| 8 | 1652411475 | 21.22550156 | 0.522626028 | 0.72660015 |
| 9 | 10648873950 | 23.08871999 | 0.549726805 | 0.747522024 |
| 10 | 62828356305 | 24.86367234 | 0.576136157 | 0.765628174 |
| 11 | 3.427E+11 | 26.56012163 | 0.601971486 | 0.781456694 |
| 12 | 1.74206E+12 | 28.18608885 | 0.627322615 | 0.795411957 |
| 13 | 8.30828E+12 | 29.74827387 | 0.652259893 | 0.807805226 |
| 14 | 3.73873E+13 | 31.25235127 | 0.676839501 | 0.818880855 |
| 15 | 1.59519E+14 | 32.70318415 | 0.701107048 | 0.828833859 |
| 16 | 6.48046E+14 | 34.1049827 | 0.725100078 | 0.837822083 |
| 17 | 2.51594E+15 | 35.4614241 | 0.748849881 | 0.845974847 |
| 18 | 9.3649E+15 | 36.77574496 | 0.772382808 | 0.853399232 |
| 19 | 3.35165E+16 | 38.05081369 | 0.795721261 | 0.860184741 |
| 20 | 1.15632E+17 | 39.28918792 | 0.818884446 | 0.866406816 |
| 21 | 3.8544E+17 | 40.49316072 | 0.84188895 | 0.872129523 |
| 22 | 1.24392E+18 | 41.66479814 | 0.864749193 | 0.877407641 |
| 23 | 3.89401E+18 | 42.80597005 | 0.887477794 | 0.882288296 |
| 24 | 1.18443E+19 | 43.91837566 | 0.910085848 | 0.88681226 |
| 25 | 3.5059E+19 | 45.00356493 | 0.932583169 | 0.891014994 |
| 26 | 1.01132E+20 | 46.0629565 | 0.954978471 | 0.89492748 |
| 27 | 2.84667E+20 | 47.09785298 | 0.977279528 | 0.898576916 |
| 28 | 7.82835E+20 | 48.10945389 | 0.999493303 | 0.901987268 |
| 29 | 2.10556E+21 | 49.09886689 | 1.021626052 | 0.905179739 |
| 30 | 5.54463E+21 | 50.06711736 | 1.043683421 | 0.908173155 |
| 31 | 1.43087E+22 | 51.01515679 | 1.065670516 | 0.910984284 |
| 32 | 3.6219E+22 | 51.94387004 | 1.087591972 | 0.913628113 |
| 33 | 8.99987E+22 | 52.85408172 | 1.109452006 | 0.916118071 |
| 34 | 2.19703E+23 | 53.74656181 | 1.131254467 | 0.918466228 |
| 35 | 5.27286E+23 | 54.62203054 | 1.153002873 | 0.920683463 |
| 36 | 1.24498E+24 | 55.48116286 | 1.174700452 | 0.9227796 |
| 37 | 2.89374E+24 | 56.32459225 | 1.196350168 | 0.924763536 |
| 38 | 6.62514E+24 | 57.1529142 | 1.217954752 | 0.926643346 |
| 39 | 1.4949E+25 | 57.96668937 | 1.239516722 | 0.928426373 |
| 40 | 3.32616E+25 | 58.76644629 | 1.261038406 | 0.930119309 |
| 41 | 7.30133E+25 | 59.55268389 | 1.282521958 | 0.931728264 |
| 42 | 1.58195E+26 | 60.32587378 | 1.303969378 | 0.933258829 |
| 43 | 3.38465E+26 | 61.08646224 | 1.325382522 | 0.934716125 |
| 44 | 7.15391E+26 | 61.8348721 | 1.346763119 | 0.936104855 |
| 45 | 1.49437E+27 | 62.57150439 | 1.368112778 | 0.937429341 |
| 46 | 3.08621E+27 | 63.29673989 | 1.389433002 | 0.938693566 |
| 47 | 6.30374E+27 | 64.01094048 | 1.410725193 | 0.9399012 |
| 48 | 1.27388E+28 | 64.71445045 | 1.431990666 | 0.941055635 |
| 49 | 2.54776E+28 | 65.40759763 | 1.45323065 | 0.942160007 |
| 50 | 5.04457E+28 | 66.09069447 | 1.4744463 | 0.943217222 |
| 51 | 9.89131E+28 | 66.76403902 | 1.495638697 | 0.944229971 |
| 52 | 1.9212E+29 | 67.42791582 | 1.516808861 | 0.945200757 |
| 53 | 3.6974E+29 | 68.08259672 | 1.537957749 | 0.946131903 |
| 54 | 7.05244E+29 | 68.72834166 | 1.559086264 | 0.947025573 |
| 55 | 1.33355E+30 | 69.36539938 | 1.580195257 | 0.947883783 |
| 56 | 2.50041E+30 | 69.99400804 | 1.60128553 | 0.948708411 |
| 57 | 4.64989E+30 | 70.61439586 | 1.622357843 | 0.949501213 |
| 58 | 8.57824E+30 | 71.22678169 | 1.643412912 | 0.95026383 |
| 59 | 1.57025E+31 | 71.83137547 | 1.664451416 | 0.950997796 |
| 60 | 2.85263E+31 | 72.42837879 | 1.685473998 | 0.951704548 |
| 61 | 5.14408E+31 | 73.01798529 | 1.706481267 | 0.952385432 |
| 62 | 9.20957E+31 | 73.60038111 | 1.7274738 | 0.953041713 |
| 63 | 1.63726E+32 | 74.17574525 | 1.748452147 | 0.953674575 |
| 64 | 2.89078E+32 | 74.74424999 | 1.769416829 | 0.954285135 |
| 65 | 5.06999E+32 | 75.30606117 | 1.79036834 | 0.95487444 |
| 66 | 8.83407E+32 | 75.86133855 | 1.811307153 | 0.955443478 |
| 67 | 1.52948E+33 | 76.41023613 | 1.832233716 | 0.955993177 |
| 68 | 2.63161E+33 | 76.95290236 | 1.853148456 | 0.956524415 |
| 69 | 4.50043E+33 | 77.48948048 | 1.874051781 | 0.957038017 |
| 70 | 7.65073E+33 | 78.02010873 | 1.894944079 | 0.957534764 |
| 71 | 1.29308E+34 | 78.54492059 | 1.915825721 | 0.958015394 |
| 72 | 2.17309E+34 | 79.06404502 | 1.936697061 | 0.958480604 |
| 73 | 3.63175E+34 | 79.57760662 | 1.957558438 | 0.958931052 |
| 74 | 6.03655E+34 | 80.08572588 | 1.978410175 | 0.959367363 |
| 75 | 9.98043E+34 | 80.58851934 | 1.999252581 | 0.959790129 |
| 76 | 1.64152E+35 | 81.08609973 | 2.020085953 | 0.960199908 |
| 77 | 2.68612E+35 | 81.57857622 | 2.040910573 | 0.960597234 |
| 78 | 4.37356E+35 | 82.06605448 | 2.061726714 | 0.960982609 |
| 79 | 7.08627E+35 | 82.54863689 | 2.082534635 | 0.961356514 |
| 80 | 1.14266E+36 | 83.02642266 | 2.103334587 | 0.961719401 |
| 81 | 1.8339E+36 | 83.49950796 | 2.124126809 | 0.962071705 |
| 82 | 2.92977E+36 | 83.96798603 | 2.14491153 | 0.962413835 |
| 83 | 4.65939E+36 | 84.43194735 | 2.165688971 | 0.962746183 |
| 84 | 7.37736E+36 | 84.89147968 | 2.186459344 | 0.963069122 |
| 85 | 1.16302E+37 | 85.34666822 | 2.207222854 | 0.963383006 |
| 86 | 1.82567E+37 | 85.7975957 | 2.227979695 | 0.963688173 |
| 87 | 2.85392E+37 | 86.24434247 | 2.248730056 | 0.963984945 |
| 88 | 4.44304E+37 | 86.68698658 | 2.269474119 | 0.964273631 |
| 89 | 6.8892E+37 | 87.12560389 | 2.290212057 | 0.964554522 |
| 90 | 1.064E+38 | 87.56026816 | 2.31094404 | 0.9648279 |
| 91 | 1.63692E+38 | 87.99105107 | 2.331670228 | 0.965094032 |
| 92 | 2.50876E+38 | 88.41802239 | 2.352390779 | 0.965353173 |
| 93 | 3.83058E+38 | 88.84124995 | 2.373105841 | 0.965605568 |
| 94 | 5.82737E+38 | 89.2607998 | 2.39381556 | 0.965851451 |
| 95 | 8.83307E+38 | 89.67673621 | 2.414520077 | 0.966091045 |
| 96 | 1.33416E+39 | 90.08912176 | 2.435219526 | 0.966324564 |
| 97 | 2.00812E+39 | 90.4980174 | 2.455914037 | 0.966552213 |
| 98 | 3.01218E+39 | 90.90348251 | 2.476603736 | 0.966774189 |
| 99 | 4.50306E+39 | 91.30557493 | 2.497288745 | 1.287457337 |
| 100 | 6.70955E+39 | 91.70435105 | 2.507672727 | 1.926043463 |
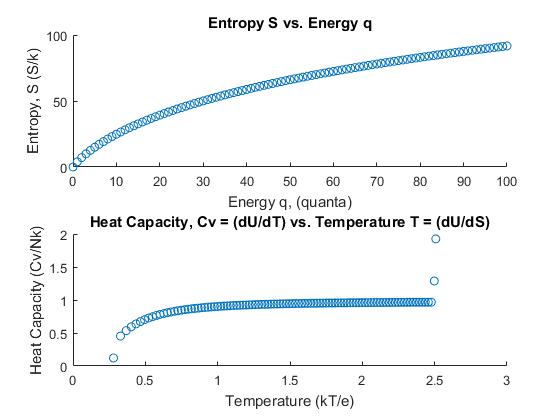
Graphing the entropy vs. energy, and the heat capacity vs. temperature gives the following:
Graphs I & II
Part II: Let units and let
. Using this in the calculation yields the following table for this Einstein solid. This “dilutes” the system and lowers the temperature:
Table II (Dimensionless Parameters):
| Energy q | Ω | S/k | kT/ε | C/Nk |
| 0 | 1 | 0 | 0 | N/A |
| 1 | 5000 | 8.517193 | 0.122388 | 0.003049 |
| 2 | 12502500 | 16.34144 | 0.131206 | 0.026553 |
| 3 | 2.08E+10 | 23.76042 | 0.137453 | 0.035575 |
| 4 | 2.61E+13 | 30.89192 | 0.14245 | 0.043342 |
| 5 | 2.61E+16 | 37.80047 | 0.146681 | 0.050387 |
| 6 | 2.18E+19 | 44.52691 | 0.150388 | 0.056922 |
| 7 | 1.56E+22 | 51.09939 | 0.153709 | 0.063064 |
| 8 | 9.74E+24 | 57.53854 | 0.156731 | 0.068885 |
| 9 | 5.42E+27 | 63.86011 | 0.159515 | 0.074436 |
| 10 | 2.72E+30 | 70.07651 | 0.162105 | 0.079755 |
| 11 | 1.24E+33 | 76.19781 | 0.164531 | 0.08487 |
| 12 | 5.16E+35 | 82.23229 | 0.166818 | 0.089804 |
| 13 | 1.99E+38 | 88.18693 | 0.168985 | 0.094575 |
| 14 | 7.13E+40 | 94.06767 | 0.171047 | 0.099198 |
| 15 | 2.38E+43 | 99.87961 | 0.173017 | 0.103687 |
| 16 | 7.47E+45 | 105.6272 | 0.174905 | 0.108052 |
| 17 | 2.2E+48 | 111.3144 | 0.176719 | 0.112303 |
| 18 | 6.14E+50 | 116.9446 | 0.178467 | 0.116448 |
| 19 | 1.62E+53 | 122.5209 | 0.180154 | 0.120494 |
| 20 | 4.07E+55 | 128.0462 | 0.181787 | 0.124447 |
| 21 | 9.73E+57 | 133.5229 | 0.183368 | 0.128314 |
| 22 | 2.22E+60 | 138.9532 | 0.184904 | 0.132098 |
| 23 | 4.85E+62 | 144.3393 | 0.186396 | 0.135806 |
| 24 | 1.02E+65 | 149.683 | 0.187849 | 0.13944 |
| 25 | 2.04E+67 | 154.9861 | 0.189265 | 0.143004 |
| 26 | 3.94E+69 | 160.2502 | 0.190646 | 0.146503 |
| 27 | 7.34E+71 | 165.4768 | 0.191995 | 0.149939 |
| 28 | 1.32E+74 | 170.6671 | 0.193314 | 0.153316 |
| 29 | 2.28E+76 | 175.8226 | 0.194604 | 0.156635 |
| 30 | 3.83E+78 | 180.9444 | 0.195868 | 0.159899 |
| 31 | 6.21E+80 | 186.0336 | 0.197106 | 0.16311 |
| 32 | 9.77E+82 | 191.0912 | 0.19832 | 0.166272 |
| 33 | 1.49E+85 | 196.1183 | 0.199512 | 0.169384 |
| 34 | 2.21E+87 | 201.1157 | 0.200682 | 0.172451 |
| 35 | 3.17E+89 | 206.0843 | 0.201831 | 0.175472 |
| 36 | 4.44E+91 | 211.025 | 0.202961 | 0.17845 |
| 37 | 6.04E+93 | 215.9384 | 0.204073 | 0.181387 |
| 38 | 8E+95 | 220.8254 | 0.205166 | 0.184283 |
| 39 | 1.03E+98 | 225.6866 | 0.206243 | 0.18714 |
| 40 | 1.3E+100 | 230.5227 | 0.207304 | 0.18996 |
| 41 | 1.6E+102 | 235.3343 | 0.208349 | 0.192743 |
| 42 | 1.9E+104 | 240.122 | 0.209379 | 0.195491 |
| 43 | 2.3E+106 | 244.8863 | 0.210395 | 0.198205 |
| 44 | 2.6E+108 | 249.6279 | 0.211397 | 0.200885 |
| 45 | 2.9E+110 | 254.3472 | 0.212386 | 0.203533 |
| 46 | 3.2E+112 | 259.0447 | 0.213363 | 0.20615 |
| 47 | 3.4E+114 | 263.7209 | 0.214327 | 0.208736 |
| 48 | 3.6E+116 | 268.3762 | 0.215279 | 0.211293 |
| 49 | 3.7E+118 | 273.0112 | 0.21622 | 0.213821 |
| 50 | 3.7E+120 | 277.6261 | 0.21715 | 0.21632 |
| 51 | 3.7E+122 | 282.2214 | 0.218069 | 0.218793 |
| 52 | 3.6E+124 | 286.7975 | 0.218978 | 0.221238 |
| 53 | 3.4E+126 | 291.3548 | 0.219877 | 0.223658 |
| 54 | 3.2E+128 | 295.8935 | 0.220766 | 0.226052 |
| 55 | 2.9E+130 | 300.4141 | 0.221646 | 0.228422 |
| 56 | 2.7E+132 | 304.9169 | 0.222517 | 0.230768 |
| 57 | 2.4E+134 | 309.4022 | 0.22338 | 0.23309 |
| 58 | 2.1E+136 | 313.8703 | 0.224233 | 0.235388 |
| 59 | 1.8E+138 | 318.3214 | 0.225079 | 0.237665 |
| 60 | 1.5E+140 | 322.756 | 0.225917 | 0.239919 |
| 61 | 1.2E+142 | 327.1743 | 0.226746 | 0.242152 |
| 62 | 1E+144 | 331.5765 | 0.227568 | 0.244363 |
| 63 | 8.1E+145 | 335.9628 | 0.228383 | 0.246555 |
| 64 | 6.4E+147 | 340.3337 | 0.229191 | 0.248725 |
| 65 | 5E+149 | 344.6892 | 0.229991 | 0.250876 |
| 66 | 3.8E+151 | 349.0296 | 0.230785 | 0.253008 |
| 67 | 2.9E+153 | 353.3553 | 0.231572 | 0.255121 |
| 68 | 2.2E+155 | 357.6663 | 0.232353 | 0.257215 |
| 69 | 1.6E+157 | 361.9629 | 0.233127 | 0.259291 |
| 70 | 1.1E+159 | 366.2453 | 0.233896 | 0.261349 |
| 71 | 8.2E+160 | 370.5137 | 0.234658 | 0.263389 |
| 72 | 5.8E+162 | 374.7683 | 0.235414 | 0.265413 |
| 73 | 4E+164 | 379.0093 | 0.236165 | 0.267419 |
| 74 | 2.7E+166 | 383.237 | 0.23691 | 0.269409 |
| 75 | 1.9E+168 | 387.4514 | 0.23765 | 0.271382 |
| 76 | 1.2E+170 | 391.6527 | 0.238384 | 0.273339 |
| 77 | 8.2E+171 | 395.8412 | 0.239113 | 0.275281 |
| 78 | 5.3E+173 | 400.0169 | 0.239837 | 0.277207 |
| 79 | 3.4E+175 | 404.1802 | 0.240556 | 0.279118 |
| 80 | 2.2E+177 | 408.331 | 0.24127 | 0.281015 |
| 81 | 1.4E+179 | 412.4696 | 0.24198 | 0.282896 |
| 82 | 8.4E+180 | 416.5962 | 0.242684 | 0.284763 |
| 83 | 5.2E+182 | 420.7108 | 0.243384 | 0.286616 |
| 84 | 3.1E+184 | 424.8136 | 0.24408 | 0.288455 |
| 85 | 1.9E+186 | 428.9048 | 0.244771 | 0.29028 |
| 86 | 1.1E+188 | 432.9845 | 0.245458 | 0.292092 |
| 87 | 6.5E+189 | 437.0529 | 0.24614 | 0.29389 |
| 88 | 3.7E+191 | 441.11 | 0.246819 | 0.295676 |
| 89 | 2.1E+193 | 445.156 | 0.247493 | 0.297448 |
| 90 | 1.2E+195 | 449.191 | 0.248164 | 0.299208 |
| 91 | 6.7E+196 | 453.2152 | 0.24883 | 0.300955 |
| 92 | 3.7E+198 | 457.2286 | 0.249493 | 0.30269 |
| 93 | 2E+200 | 461.2315 | 0.250152 | 0.304413 |
| 94 | 1.1E+202 | 465.2238 | 0.250807 | 0.306124 |
| 95 | 5.9E+203 | 469.2057 | 0.251458 | 0.307823 |
| 96 | 3.2E+205 | 473.1774 | 0.252106 | 0.30951 |
| 97 | 1.7E+207 | 477.1389 | 0.252751 | 0.311187 |
| 98 | 8.6E+208 | 481.0903 | 0.253392 | 0.312851 |
| 99 | 4.4E+210 | 485.0318 | 0.254029 | 0.418439 |
| 100 | 2.3E+212 | 488.9634 | 0.254345 | 0.628243 |
Thus the graphs of the entropy vs. energy and heat capacity vs. temperature follow:
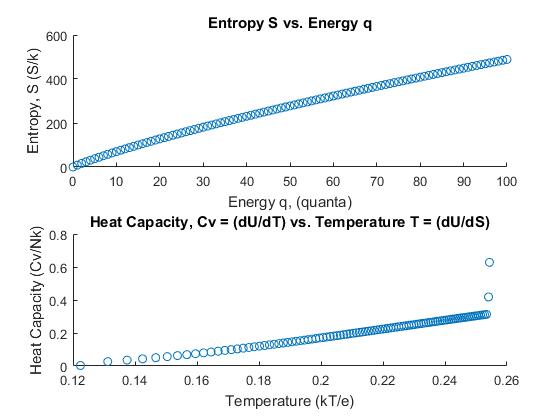
Figure 2. Graphs III and IV.
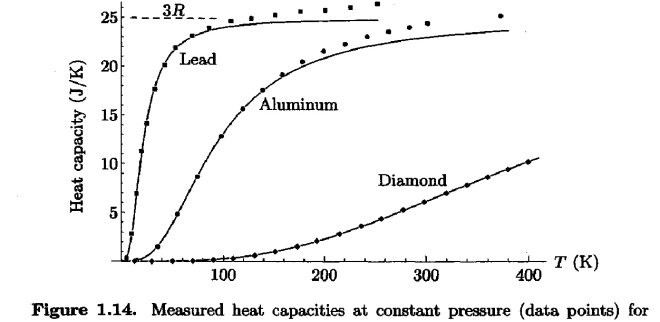
Figure 3. (Figure 1.14 of Schroeder’s Thermal Physics) Heat Capacity curves for Lead (Pb), Aluminum (Al), and Diamond, respectively as a function of temperature in Kelvin.
Graph II shows the prediction for heat capacity as a function of temperature of an Einstein solid for which there are 100 units of energy and 50 oscillators. The data exhibits a trend that appears to reach an asymptote quickly, then when the temperature reaches T ≈ 2.5, there is a sudden increase in the value of the heat capacity. The approach to determining the final data points was switched from a central difference approximation to a backward difference approximation of the last two entries corresponding to energies q = 99 and q = 100 units. If we ignore the last two, the curve approaches an asymptote at CV = 1. However, the graphs produced are of the dimensionless quantities involved. The overall curve appears to be logarithmic and resembles the heat capacity curve for lead. The initial increase is almost immediate and its slope appears to be slightly less than lead but greater than aluminum.
Graphs III and IV show the prediction for heat capacity in terms of temperature of an Einstein solid for which the energy is the same, but the number of oscillators is now 5000. The temperature has been reduced and the heat capacity vs. temperature yields a graph that shows a trendline that appears linear. Comparing to Figure 3(Fig. 1.14 in the text), this graph resembles the heat capacity curve for diamond. In Figure 3, the diamond curve is linear throughout. The only discrepancies among Graph IV and Figure 3 are the final two data points in Graph IV. Again, a backward difference approximation was used to determine the final data points for this Einstein solid as well. The value for the constant ε was determined by finding the quotient of the entropy and temperature columns and taking the average value of ε for each energy.
This was the numerical analysis of an Einstein solid’s temperature, energy, entropy, and heat capacity. In the next post, I shall discuss the analytical version of this analysis.
